The Significance of Humidity Control in GMP Compliant Production
GMP compliance in pharmaceutical manufacturing requires that any process, person, environment, or equipment with direct impact on the quality and safety of the product being produced must operate within specified limits.
These specified limits should be under the direct control of the manufacturing team, with countermeasures available in the event of a problem. In addition, any other part of the production or storage processes that have an indirect impact must also be assessed for possible risk impact.
Humidity may not seem like an obvious cause of problems, or something that could even result in production being non-compliant, but this can and does occur. Just because one does not see an issue doesn’t mean there isn’t a problem. Many phenomena are influenced by the relative humidity level, and they can cause production processes to be less efficient, less predictable, and more prone to producing products that don’t meet specification.
Some common issues that can arise from poor humidity control are:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Figure 1 shows the influence on storage of a variety of materials as relative humidity changes. Storage in this sense is not just restricted to warehousing, but also applies to any material, object, or equipment that remains stationary in a fixed location for extended periods. So, think of this also in the context of equipment and fittings within a pharmaceutical manufacturing plant.
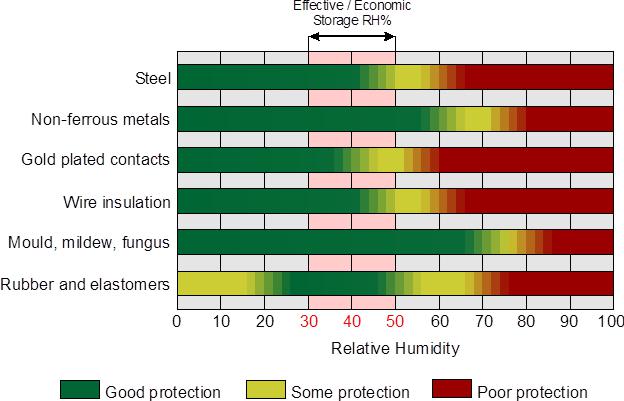
Figure 1: Effect of RH% on materials in static locations.
It is important to note the relationship between relative humidity (RH%) and air temperature, as variations in temperature also will affect the RH%. For example, a sufficiently large drop in air temperature can result in water vapor condensing out. Alternatively, cold surfaces will cause condensation to form if the surface temperature drops below the dew point of the air around it.
Humidity in Manufacturing Areas
In pharmaceutical manufacturing, quality and consistency are key. Consider the following typical conditions found in areas related to solid dosage production:
| Production area | Temperature | Humidity |
| Weighing, Mixing | 68 to 72°F (20 to 22°C) | 35 to 40% RH |
| Compression | 68°F (20°C) | 25 to 35% RH |
| Pan coating | 53 to 203°F (12 to 95°C) | 10 to 70% RH |
| Filling and Packing | 68°F (20°C) | 10 to 35% RH |
| Storage | 68 to 77°F (20 to 25°C) | 45% RH |
Figure 2: Typical conditions in solid dosage production.
From Figure 2, if the RH% levels therein are correctly maintained for the designated process and surrounding areas, then no humidity-related issues are likely to occur, either to the material produced or the production equipment itself.
Of course, the actual required RH% values for any given process are dependent upon many factors, including:
- The materials being used and their sorption isotherms, hygroscopicity, and sensitivity to water.
- The manufacturing space itself: volume, building insulation, airflow control, number of openings, geographic location, climate, etc.
- SOPs and operator behavior: cleaning cycles, procedures for material transfers, etc.
- GMP specified values which may be influenced by other factors.
Often, issues can arise when there has been some change made to the manufacturing conditions, such as a new product formulation, the use of different fillers, or adding staff on the plant floor. Introducing a few extra tons of materials or adding additional personnel in the space will alter the load on the air handling system and result in humidity issues.
Therefore, in all areas of production, humidity levels always should be carefully considered, not just to suit the material being produced, but also to take into account any other effects arising from unexpected RH% levels that could occur and potentially disrupt production.
So, the questions that pharmaceutical manufacturers must ask themselves are:
- Do the relative humidity levels within my facility exceed GMP specified values at any time?
- If they do, for how long? What is the recovery time to return to the specified value?
- What is the impact of this excursion from the specified value, and do I need to react to this?
- Is recovery time fixed or variable? For example, with effective humidity control the recovery will be quicker in dry winter months or longer during wet, humid periods.
Bearing all of the preceding points in mind, take a moment to consider if you are experiencing moisture related production or quality problems in production areas. Issues may arise based on a number of factors:
- The flow of API and excipients is not as expected; it could be either too fast or too slow depending upon how moisture affects the materials.
- The angle of repose in powder or granulate (Figure 3) samples is not as expected. If a small amount of available water is able to bridge the gaps between particles, electrostatic attraction of the water to surfaces will alter the angle of repose, another sign that flow characteristics may have changed.
- Do you experience any variations when weighing batches of raw materials or finished product that could be caused by fluctuations in water content? This can be determined by referring to the sorption isotherms of the materials being weighed.
- Are there signs of clogging, caking, or other obstructions in silos, bins, pipework, and process equipment? Does pneumatic transport take longer than expected? Is there additional noise and vibration that might be attributed to blockages? It’s also important to remember that the increased air pressure of pneumatic transport systems will affect the dew point of the air within the system, and condensation could occur at higher than expected temperatures.
- When using moisture-permeable containers for packaging, such as plastic bags and semi-rigid, low-density polyethylene (LDPE) pouches for large volume parenterals (LVPs), and LDPE ampoules, bottles, and vials, due consideration should be given to the stability of the contents under high humidity conditions.
- Moisture may have an undesirable effect on chemical stability (e.g. some antibiotics may undergo hydrolysis) and physical stability (e.g. dissolution rate may change). Drugs with functional groups such as esters, amides, lactones, or lactams may be susceptible to hydrolytic degradation, which is a commonly encountered mode of drug degradation. Many polymers that are susceptible to hydrolysis, for example, the polyesters PLA and PLG degrade by random hydrolysis that takes place homogeneously throughout the bulk of the polymer device.
- Consider the impact of moisture in bulk materials and finished product.

Figure 3: The angle of repose of a powder or granulate is the steepest angle of descent or dip relative to the horizontal plane to which a material can be piled without slumping. Often a measurement is taken in pre-production as a quality check to ensure material is flowing as it should.
Even when taking all of these factors into account, more radical action might be needed. It may be appropriate to go right back to the start and ask, “How was the RH% level for production and storage processes determined in the R&D phase?”
In some cases, the environmental conditions for producing a new product are adopted from a similar, existing product already in production based on the assumptions that:
- These conditions work and are proven, and there does not appear to be any significant dissimilarities between the two products.
- Particularly in cases where an existing manufacturing area is being repurposed for a new product, the available HVAC system can produce those conditions.
Storage and Warehousing
It is important to consider a pharmaceutical product’s exposure to humidity throughout the entire production process. In storage and warehousing, deviations from the desired temperature and humidity conditions must be minimized, controlled, and documented.
Unfortunately, temperature and humidity excursions are almost inevitable, but automated control of HVAC and humidity control systems will improve response and recovery, and also can provide historical and trend data to track these excursions.
Although minor deviations probably have no significant impact, it’s important to consider the effects of temperature or humidity deviations on every item held in storage. This can be a daunting task, as it is not unusual for warehouses to contain hundreds or even thousands of different inventory items.
It also is worth considering that new items will be added in the future, which will require some form of assessment. The impact assessment, therefore, has the potential to become an enormous task.
It is easier to ensure temperature and humidity are controlled within defined limits (e.g. 72°F/22°C at 50% RH) supported by automatically generated logs. Keep in mind that varying air handling strategies may be necessary based on the location and local environment of each storage facility.
Psychrometrics and Humidity Control
There are different ways to control humidity, or more correctly, to attempt to control humidity.
One way is using outdoor air for ventilation. With this method, the ventilation air must have a lower moisture content than the air within the building to be effective, and is therefore at the mercy of changing weather and seasonal conditions.
Thus, for the most part, we should ignore using untreated outdoor air because of its variability. Instead, let’s review more effective ways to treat the air either entering or already within the building. These methods include:
- Heating – This application will lower the relative humidity, but absolute humidity remains unchanged. There still is the same mass of water vapor and the dew point is unchanged. This might be a reasonable humidity control strategy for heating the area for comfort, but in energy terms it can be relatively expensive.
- Cooling – Using cooling coils to reduce the air temperature below its dew point is more common. This method will lower the RH% after cold air is re-heated, and it also will reduce absolute humidity. However, efficiency falls significantly as air temperatures fall below 50°F (10°C). In addition, the inevitable condensation that forms on the cooling coils can become a maintenance issue if they are prone to corrosion. Finally, the wet conditions are a good breeding ground for bacteria and mold, which are not wanted anywhere near pharmaceutical production.
- Desiccant Dehumidification (DH) – This method reduces both relative and absolute humidity, and also reduces dew point, while not being temperature sensitive, with an operating range between 100°F to -40°F (40°C to -40°C). The system allows for lower airflows when compared to other methods of air treatment, resulting in energy savings. This form of precise humidity control also is very flexible for multiple energy sources (e.g. gas, steam, LPHW, etc.), so available utilities and waste heat can be used. Additionally, the system runs dry, which reduces the possibility of microbial growth and maintenance arising from wet conditions, and also can translate to longer equipment life. Furthermore, this form of humidity control can dry down to a -94°F (-70°C) dew point, which may be required for sensitive APIs.
Conclusion
Once a target RH range is specified for a facility, those set points should be maintained for quality and consistency in production. Implementing an effective humidity control strategy in pharmaceutical production and storage will ensure year-round GMP compliance and enhance product safety.
About the Author
Martin Ginty is Global Pharmaceutical Industry Manager for Munters, a provider of air treatment solutions for humidity and climate control. He has more than 20 years of experience in implementing national and international level automation and HVAC projects.
